Lobby against alcohol cancer risk: perspective from the European Parliament corridors
Leaflet distributed by Eurocare before the ENVI committe vote
EU policy context
In February 2021, the European Commission unveiled its ambitious Europe's Beating Cancer Plan: a bold initiative aimed at leaving no stone unturned in the fight against cancer. As part of this comprehensive plan, the Commission introduced a roadmap for implementation, encompassing actions like mandatory ingredient labeling and nutritional declarations on alcoholic beverage products, along with a legislative proposal for health warnings on such products. The broader effort to tackle non-communicable diseases (NCDs) is detailed in another Commission initiative known as Healthier together – EU non-communicable diseases (NCD) initiative.
The European Parliament's contentious debate on alcohol and NCDs
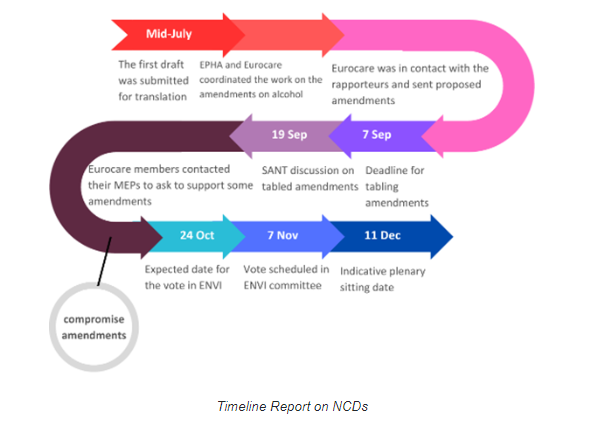
In the summer of this year, members of the European Parliament, specifically those belonging to the health sub-committee (SANT), initiated a report that aimed to address risk factors contributing to NCDs, including recommendations for the European Commission. The SANT sub-committee is a part of the Environment, Public Health, and Food Safety (ENVI) Committee.
The process of creating this report was an arduous one, with Members of the European Parliament submitting over 500 amendments, including 12 addressing alcohol-related matters. Eventually, political groups, along with rapporteur Erik Poulsen (Renew/Denmark), reached a consensus on a reduced set of compromise amendments. It is worth noting that debates surrounding alcohol have consistently ignited substantial disagreements in Parliament. Notably, the alcohol section emerged as a focal point of contention during deliberations on the Cancer plan.
However, on November 7, 2023, the ENVI Committee voted on the compromise amendments, yielding results that were far from satisfactory. Notably, a reference to health warning labeling – as a recommendation to the EU executive for improving the information to consumers – disappeared from the compromise amendments entirely. Additionally, the references to "alcohol consumption" were removed from critical sections of the text, specifically those that listed factors and determinants substantially increasing the risk of NCDs. The text maintains only a mention of the “harmful use of alcohol.” This, however, poses a concern, as scientific evidence as well as the WHO and IARC consistently indicate that any level of alcohol consumption, even in modest quantities, heightens the susceptibility to NCDs.
Eurocare advocacy efforts
 This process underwent scrutiny by Eurocare alongside our members and partners. From the onset, Eurocare actively engaged with leader MEPs to advocate for proposals that aligned with scientific evidence and the public's demands. From June onwards, the Eurocare Secretariat kept its members and partners informed through our regular Alcohol Policy Coordination meetings (APCMs). During these, we explained the policy process and the timeline, the analysis of the amendments related to alcohol in September, and the compromise amendments in October and November. Eurocare and its members as well as EPHA and Movendi had a common view that the terminology of ‘moderate and responsible drinking information’ or ‘harmful consumption of alcohol’ was scientifically inaccurate and deliberately unclear, and we agreed to do all we could to ensure that this would not be the wording in the final report. Eurocare prepared a model letter which was translated and sent to many MEPs. Our combined commitment in the effort is testament to the importance the health community placed on this work. We would like to thank everyone who took the time to engage with their MEPs for their exceptional work on this file.
This process underwent scrutiny by Eurocare alongside our members and partners. From the onset, Eurocare actively engaged with leader MEPs to advocate for proposals that aligned with scientific evidence and the public's demands. From June onwards, the Eurocare Secretariat kept its members and partners informed through our regular Alcohol Policy Coordination meetings (APCMs). During these, we explained the policy process and the timeline, the analysis of the amendments related to alcohol in September, and the compromise amendments in October and November. Eurocare and its members as well as EPHA and Movendi had a common view that the terminology of ‘moderate and responsible drinking information’ or ‘harmful consumption of alcohol’ was scientifically inaccurate and deliberately unclear, and we agreed to do all we could to ensure that this would not be the wording in the final report. Eurocare prepared a model letter which was translated and sent to many MEPs. Our combined commitment in the effort is testament to the importance the health community placed on this work. We would like to thank everyone who took the time to engage with their MEPs for their exceptional work on this file.

Unprecedented role from the WHO and IARC
The importance of accurate wording in the report was emphasized by the unprecedented action of the World Health Organization (WHO) and the International Agency for Research on Cancer (IARC) which, ahead of the vote in the ENVI Committee on the 7th of November, issued a joint statement emphasizing the crucial connection between any alcohol consumption and cancer incidence and mortality. They called for a clear acknowledgment of the role of alcohol consumption in cancer, devoid of any qualifiers or misleading adjectives. Expressing their concerns, the Regional Director of WHO/Europe and the Director of the IARC addressed MEPs, the President of the European Parliament, Roberta Metsola, and the Commissioner for Health and Food Safety, Stella Kyriakides. The letters clearly highlighted the worrisome scientific inaccuracy of terms like ‘moderate and responsible drinking information’ and ‘harmful consumption of alcohol’ in the context of cancer prevention. In addition, the removal of the European Commission's commitment to introduce cancer-specific health warning labels was highlighted. Finally, while recognizing the non-legislative nature of the report, the WHO expressed strong concerns about “[…] the proliferation of this terminology within the European Parliament, along with the outlined absence of commitments to health warnings in recent documents […].
What about the media?
Eurocare had several meetings with a number of journalists, including Euractiv and the New York Times, and we were pleased that our efforts resulted in a strong article on Euractiv, shedding light on the WHO’s concerns about the NCD report’s language regarding alcohol, and stressing the dangers of the situation on the eve of the vote.
What happened during the vote on the 7th of November?

Ahead of the vote, Eurocare and the European Association for the Study of the Liver (EASL) issued a press release urging MEPs to make choices consistent with WHO recommendations and scientific evidence that not drinking alcohol is better for cancer prevention. Just before the ENVI committee vote, Eurocare staff and partners distributed the Alcohol and Cancer in the European Union - A Call to Action WHO & IARC statement, and a flyer detailing which compromise amendments to reject and support for the best possible outcome. Regrettably, despite our efforts, Bloc Vote 2 (Paragraphs 3, 6 and 7) – which favors the ambiguous position on alcohol consumption and cancer which has been consistently promoted by the alcohol industry – was approved.
It appears that some MEPs continue to prioritize the interests of the alcohol industry ahead of the scientific evidence base and the public interest. In this case, this results in an unclear and inaccurate message to European citizens. Eurocare’s view is that the actions of the rapporteur, Mr Erik Poulsen (Renew/ Denmark), do not continue the work of his predecessor, the late Mme. Veronique Trillet-Lenoir (Renew/ France), who led the process which produced the BECA report; Strengthening Europe in the Fight Against Cancer.

Eurocare and Rebecka Öberg (IOGT-NTO) before the ENVI Committee vote
Mobilizing for the Future: Eurocare's campaign and the upcoming December plenary vote
It is imperative that civil society redoubles its advocacy efforts as we approach the plenary vote scheduled for 11th December. |
The implications of this vote are profound. The WHO and IARC intervention makes clear that the report as passed on 7th November is not consistent with scientific evidence, despite our campaign. Looking ahead, the next step for the report's adoption involves the plenary vote in December 2023. Eurocare intends to rally its networks and partners to garner sufficient support for tabling amendments before the plenary vote. The support of 71 members of the parliament will be necessary for the text to still have a chance of being amended. Eurocare will continue to work closely with its members, as well as other partners including IOGT NTO, ECL, Movendi, EPHA ECCO and others. Eurocare already engaged in discussions with several supportive MEPs, including Sara Cerdas (S&D/Portugal), MEP Allessandra Moretti (S&D/Italy) and MEP Michel Rivasi (Greens/France) and we will keep you informed.

Eurocare team with MEPs Alessandra Moretti (S&D/Italy), Marie Arena (S&D/Belgium) & Sara Cerdas (S&D/Portugal)


